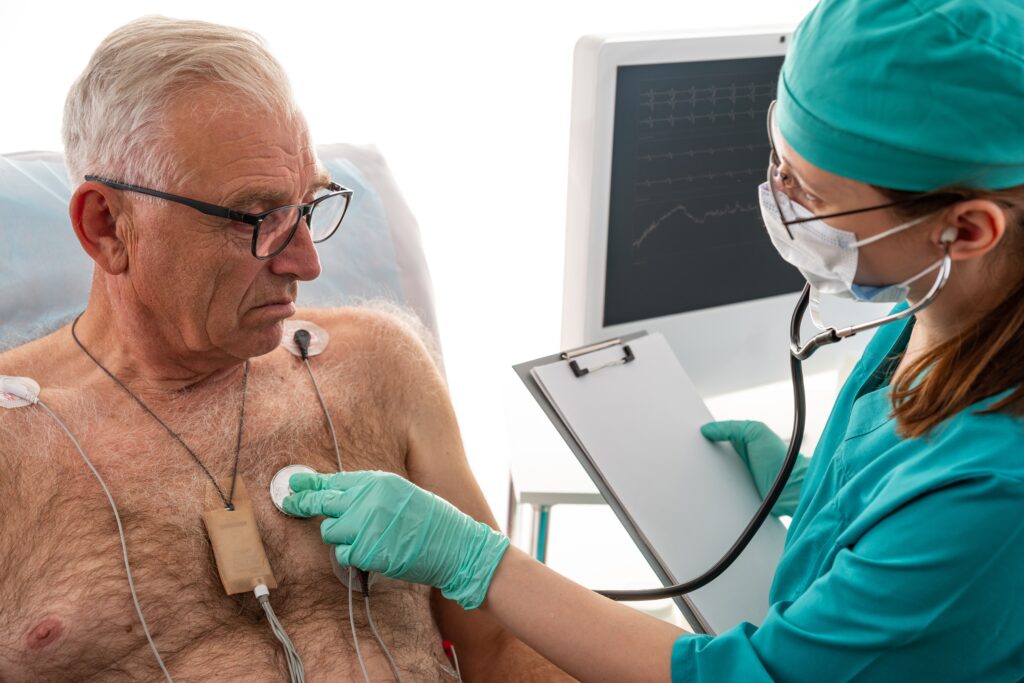
If you or a loved one has been injured or killed by a defective Medtronic Heartware Ventricular Assist Device (HVAD) System, you need experienced, expert defective medical device lawyers like Nadrich Accident Injury Lawyers to fight for your right to obtain all of the financial compensation you are entitled to under California law.
California law allows those injured by defective medical devices to be financially compensated for their injuries by the manufacturers of the defective devices. At least 13 recalls have been issued regarding the Medtronic HVAD System and its defective components.
The lawyers at Nadrich Accident Injury Lawyers don’t charge a fee for their representation until and unless they obtain a financial recovery for you. This means you will never owe us a penny out of your own pocket, so you have nothing to lose by calling us.
Call us today at 800-718-4658 for a free consultation, or text us from this page, if you or a loved one was injured or killed by a Medtronic HVAD System. You may be entitled to a financial recovery.
Medtronic HVAD System Recalls
The Medtronic HVAD System and its components have been recalled over a dozen times. Medtronic HVAD System recalls and notifications include:
April 2022
The FDA, in April 2022, alerted healthcare providers that a welding defect in the Medtronic HVAD System’s internal pump may cause the pump to malfunction, leading to pump thrombosis.
Pump thrombosis is a major ventricular assist device malfunction involving the blockage of flow of blood in a blood vessel. Pump thrombosis can necessitate surgery and can be fatal.
Patients who developed pump thrombosis from the Medtronic HVAC System, according to the FDA, experienced one or more of the following symptoms:
- Grinding sound
- Elevated lactate dehydrogenase
- Transient power spikes on the log files and High Watt alarms
- Dizziness or lightheadedness
- Low motor speed resulting in low perfusion
Medtronic received three complaints about this issue and all three patients were given pump exchanges. Two of the three patients died after the pump exchanges.
June/August 2022
The FDA, in June 2022, alerted healthcare providers that Medtronic stopped selling and distributing the HVAD System because:
- The internal pump is associated with an increased risk of mortality and neurological adverse events
- The internal pump may stop, and when it stops, it may delay restarting or fail to restart
Both of these issues, according to the FDA, may lead to:
- Death
- Stroke
- Worsening heart failure
- Heart attack
- Hospitalization
- A need for additional procedures
Medtronic has reported that over 100 complaints exist regarding the HVAD internal pump failing to restart or experiencing a delay in restarting, leading to 13 pump removals and 14 deaths.
The FDA, in August 2022, upgraded the recall based on these issues to a Class I recall, the most serious recall type.
May 2021
The FDA, in May 2021, announced a recall of the HVAD System’s instructions for use and patient manual. The reason for the recall was that serious patient harm, including death, could result if:
- The carrying case breaks and the driveline pulls out of the controller as it drops
- The driveline disconnections from backwards driveline cover orientation
- A controller exchange is performed unnecessarily due to confusion of start-up behavior as a “red alarm” battery failure
The recall updated the instructions for use and patient manual to:
- Give information about the carrying cases’ lifespan and how to safely wear and clean the Convertible Patient Pack
- Inform patients to keep the driveline cover on when disconnecting and reconnecting the driveline during controller exchanges
- Make it clear that the expected power-up sequence causes alarm indicator LEDs and battery LEDs to turn red for 2.5 seconds while the controller LCD displays a power-on message
One death and 64 injuries were reported to the FDA regarding these issues.
April 2021
The FDA, in April 2021, announced a recall of the HVAD System’s data cables, battery cables, controller 2.0 ports and adapter cables due to a risk of damage to controller ports.
The recall occurred because Medtronic identified a risk of wear and tear of connector plugs that could cause damage to controller port pins, such as bent pins. Damaged controller ports could lead to a partial or full stop of the pump, which could cause:
- Death
- Heart attack
- Hospitalization
- Loss of consciousness
This issue saw 12 deaths and 8 injuries reported to the FDA.
May 2020
The FDA, in May 2020, announced a recall of the HVAD System’s pump outflow graft and outflow graft strain relief due to a risk of tears and breaks during setup.
Use of the affected products might cause serious harm to patients, according to the FDA, including:
- Death
- Additional medical procedures
- Fluid buildup around the heart
- Bleeding
- Loss of consciousness
- Dizziness
Medtronic received 92 complaints regarding this issue.
May 2018
The FDA, in June 2020, announced a recall of the HVAD System after Medtronic, in May 2018, sent a letter to healthcare providers warning about the potential for interruption of the electrical connection between the HVAD System’s power source and the HVAD controller. The issue, according to the letter, was caused by oxidation of connecting surfaces.
Interruption of the HVAD controller may cause the pump to stop working, leading to:
- Death
- Nausea
- Anxiety
- Dizziness
- Weakness
April 2017
The FDA, in April 2017, announced a Medtronic HVAD System controller recall because of the possibility that the device could experience power and data failures, battery failure, or premature wear of the alignment guides which could prevent treatment.
October 2016
The FDA, in October 2016, announced two separate recalls regarding the HVAD System.
The first recall was issued because of an issue with the driveline connector that might let water or other material enter the heart pump, causing a device malfunction which could lead to serious injury or death.
The second recall was issued because a loose connector could cause the rear portion of the pump’s driveline connector to separate from the front part of the connector, letting moisture into the device which could cause the pump to stop, possibly leading to serious injury or death.
August 2015
The FDA, in August 2015, warned that the HVAD System was linked to a large number of strokes, as well as bleeding complications.
June 2015
The FDA, in June 2015, announced an HVAD System recall because the power supply connector ports might wear down over time, preventing the device controller from connecting to the system, causing the pump to stop, possibly leading to serious injury or death.
The recall was issued after at least 33 reports of device malfunction.
December 2013
The FDA, in December 13, announced an HVAD System recall because the driveline connector locking mechanism might fail to engage, causing the device to fail and stop pumping, leading to serious injury or death.
The FDA later classified this recall as a Class I recall in April 2014.
Why Choose Nadrich Accident Injury Lawyers?
Companies who manufacture medical devices are duty-bound to ensure their devices are safe. When companies put profit over people, innocent people get killed or injured. Often, it’s not until major litigation presents a threat that companies decide to recall dangerous and defective products.
At Nadrich Accident Injury Lawyers, we fight for you against powerful, large corporations which cause harm to innocent people. Our experienced, expert defective medical device attorneys have been obtaining financial compensation for clients since 1990. We have held corporations responsible for their defective products. We have represented clients after countless product recalls and have built a reputation for success in handling cases such as these.
When we evaluate your Medtronic HVAD case, we will determine if you or a loved one were using the HVAD system under the direction of a healthcare provider and if you or a loved one suffered any of the following injuries:
- Death
- Neurological dysfunction
- Worsening of heart failure symptoms
- Weakness
- Dizziness
- Stroke
- Bleeding
- Nausea
- Anxiety
- Sepsis
- Loss of consciousness
Heart patients can be very weak, so lawsuits involving the HVAD System can be very complicated. Because of this patients and those they love who think they might have a valid lawsuit should consider talking with a lawyer who is experienced in complex defective medical device cases.
The battle-tested attorneys at Nadrich Accident Injury Lawyers will be able to tell you if you can file a lawsuit on time. There is a time limit to file a lawsuit regarding the HVAD System, and this time limit is known as a statute of limitations. You might never be able to obtain financial compensation for your injuries if this deadline passes. That’s why it’s vital for you to consult with us as soon as possible about a possible HVAD System lawsuit against Medtronic. Let our highly compassionate and skilled lawyers do what’s necessary to protect your interests and rights.
Types Of Compensation In A Medtronic HVAD System Lawsuit
You might qualify for numerous types of financial compensation in a Medtronic HVAD System lawsuit, including:
Past and future medical bills: You might be compensated for past medical costs. You might also be compensated for any future medical procedures or treatments which may be necessitated by your use of the Medtronic HVAD System.
Loss of wages: You might be compensated for any income you lose because of being in the hospital for your HVAD System injuries. You might also be compensated for any income you lose in the future if your HVAD System injuries cause you to be permanently unable to work.
Mental and physical pain and suffering: Defective HVAD Systems may cause painful injuries or cause you to undergo painful medical procedures, and it can also mentally traumatize you to realize you’ve been hurt by a medical device manufacturer’s negligence. You might be compensated financially for emotional suffering and physical pain.
Punitive damages, if appropriate: If a court decides that Medtronic was selling HVAD Systems despite knowing they were dangerous, punitive damages may be awarded to punish Medtronic and deter other medical device manufacturers from behaving similarly.
Wrongful death: If you lost a loved one to a defective HVAD System, you may be entitled to compensation for wrongful death damages such as any financial support your loved one would have contributed, funeral and burial expenses, loss of love and companionship, and more.
How Can Nadrich Accident Injury Lawyers Help?
You need a qualified law firm to represent you in a Medtronic HVAD lawsuit. You will only feel good about the team you choose and how effective they will be long-term if they are qualified.
We have been representing clients in similar lawsuits for over 30 years. We are familiar with all applicable state and federal regulations and can provide you with assistance immediately.
One of the most important things you can do in a Medtronic Heartware lawsuit is have a lawyer by your side. You don’t want to settle for less and this is where our legal expertise can help you. We can teach you about how your case must be managed and what steps need to be taken as your case moves forward.
Services we can provide you include evidence collection, managing trials, filing legal claims and handling the settlement process. We will handle everything about the legal process with care for you, including how you are represented in court.
We are experts in handling defective medical device claims and because of this we will provide you with ample legal resources and we will do everything according to your requirements.
Our attorneys are widely regarded as some of the finest in California with tons of experience in defective medical device cases. We will always give it our all to help you. We are passionate, focused and will fight for your rights immediately.
To learn more about how we can help you in a Medtronic Heartware HVAD lawsuit, call us today at 800-718-4658 or text us from this page for a free consultation.