Medtronic has recalled the HawkOne and TurboHawk Plus directional atherectomy systems because their catheter tips may break off or separate inside arteries, leading to serious injuries.
When you go to a medical provider for a medical treatment, you expect that treatment to improve your health, not make it worse because a defective medical device was used. Being injured by a defective medical device could leave you unable to work and in pain with huge medical bills to pay.
Luckily, California law allows those injured by defective medical devices to recover financial compensation for their injuries from the manufacturers of the devices. However, medical device manufacturers won’t simply compensate you for your injuries if you ask them nicely.
If you’ve been injured by a Medtronic HawkOne or TurboHawk Plus directional atherectomy system, you’ll need strong, experienced lawyers like Nadrich Accident Injury Lawyers to prove that your injury was caused by a defective medical device, fight for your legal rights and force Medtronic to pay you what you deserve and are legally entitled to for your injury.
Our battle-tested, expert defective medical device lawyers have recovered hundreds of millions of dollars for clients since 1990 and only charge a percentage of any recovery we obtain for our clients. This fee, known as a contingency fee, is never charged unless and until we win your case and obtain financial compensation for you.
Call us today at 800-718-4658 for a free consultation, or text us from this page, if you or a loved one was injured by a HawkOne or TurboHawk Plus directional atherectomy system.
HawkOne Recall And TurboHawk Plus Recall Reasons
The HawkOne and TurboHawk Plus directional atherectomy systems are medical devices which are used to remove peripheral artery blockages and improve blood flow during medical procedures. The devices consist of a cutter driver and a catheter.
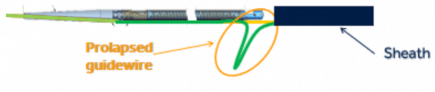
The devices have been recalled because the guide wire inside the catheter can move downward or prolapse when force is applied during a procedure. This may cause the catheter tip to separate or break off, leading to serious injuries including:
- Arterial dissection, where the inside wall of an artery is torn
- Arterial rupture, where an artery breaks
- Ischemia, where a blocked artery causes blood flow to a body part to be decreased
- Blood vessel complications which may necessitate surgery and additional procedures to remove the tip of the device
There have been at least 55 reports of injuries relating to the devices reported to the FDA, and over 95,000 devices have been recalled.
Have The HawkOne And TurboHawk Plus Devices Been Removed From The Market?
No. The recall actions taken by Medtronic have consisted of sending urgent medical device notice letters to affected customers. The letters reiterate the existing precautions and warnings in the devices’ instructions for use related to the risk of guide wire prolapse and tip damage. The letters inform affected customers of reports of tip damage during use of the devices which have resulted in tip detachment and embolization.
The letters inform affected customers that no updates will be applied to the devices at this time, patients should continue to receive monitoring as per normal procedures, no actions are necessary for patients who previously underwent a procedure utilizing the devices, and no product disposals or retrievals have been requested by Medtronic.
The letters request that affected customers take the following actions:
- Share the letter with anyone who needs to read it in their organization or in organizations that the devices have been transferred to
- Review the devices’ instructions for use before using them, noting the precautions and warnings in the letter
- Complete and email an customer confirmation form enclosed in the letter to Medtronic
Do I Have A Valid HawkOne Lawsuit Or TurboHawk Plus Lawsuit?
You can file a HawkOne recall lawsuit or a TurboHawk Plus recall lawsuit if you or a loved one has been injured by one of these devices.
California law holds medical device manufacturers strictly liable for any injuries caused by defective medical devices. This means the manufacturers are financially liable for injuries their defective devices cause regardless if those defects are the result of negligence by the manufacturers.
There are two types of product defects: manufacturing defects and design defects.
Manufacturing defects are defects which occur because something went wrong during a product’s manufacturing process. Medical devices with manufacturing defects are different than the the manufacturer’s specifications or design and other typical units in the same line of product.
A lawsuit for strict liability: manufacturing defect will be successful if it can prove the following:
- A defendant made, sold or distributed a product
- The product had a manufacturing defect when it left their possession
- A plaintiff was harmed
- The manufacturing defect substantially caused the harm to the plaintiff
Products with design defects, however, didn’t have anything wrong happen during the manufacturing process. They are unreasonably dangerous simply due to the design of the product itself. The design itself is defective.
A lawsuit for strict liability: design defect must prove the following to be successful:
- A defendant made, sold or distributed a product
- The product didn’t perform as safely as an ordinary consumer would expect it to when used as instructed or in a reasonably foreseeable manner
- A plaintiff was harmed
- The product not performing safely substantially caused the harm to the plaintiff
California juries are also asked to consider the following when evaluating design defect claims:
- How badly someone can be hurt by using the product
- How likely someone is to be hurt by the product
- How feasible alternative, safer designs were when the product was made
- How much alternative designs cost
- Alternative designs’ disadvantages
Whether the HawkOne and TurboHawk Plus devices contain a manufacturing defect or a design defect, the expert product liability lawyers at Nadrich Accident Injury Lawyers have been handling manufacturing defect and design defect cases for decades. We have recovered countless settlements for victims of defective medical devices such as Philips CPAP machines and defective hernia mesh devices, as well as other blood vessel catheters such as Penumbra Jet 7 Catheters.
Do I Need A Defective Medical Device Lawyer?

You need experienced, expert defective medical device attorneys like Nadrich Accident Injury Lawyers by your side if you want to recover the largest possible settlement for your HawkOne or TurboHawk Plus injuries.
Medtronic is a huge corporation. They reported $30.117 billion in total revenue and $93.083 billion in total assets for the fiscal year ending April 30, 2021. A huge corporation like this can afford very expensive, very skilled lawyers.
A huge corporation is always going to prioritize its own profit over the health of a single person. Therefore, if you simply ask Medtronic nicely to pay for your medical bills after a HawkOne or TurboHawk Plus device injured you, you are never going to recover the compensation you deserve and are entitled to under California law. Medtronic won’t pay you for your injuries and will dare you to take them to court without a lawyer, knowing you can’t win.
Our HawkOne lawyers, however, have been successfully taking on huge corporations like Medtronic for over 30 years. Our TurboHawk Plus lawyers relish the role of David taking on Goliath, and are very good at playing this role. In fact, most of our cases end up settling out of court because of our long, established track record of success. Companies see our names on a lawsuit and typically decide to settle out of court to avoid expensive legal fees since they know they can’t win against us.
The process of filing a medical device injury claim is a long, complicated and time-consuming process, with many elements that a layman will simply not know how to do properly. Going through this entire process alone is the last thing you should be doing while you are recovering from your injuries. You should let experienced, knowledgeable lawyers like Nadrich Accident Injury Lawyers take care of every aspect of this process for you while you simply focus on recovering from your injuries.
Call our HawkOne recall lawyers at 800-718-4658 today for a free consultation. Our TurboHawk Plus recall lawyers will handle every step of your lawsuit for you so you can simply get better from your injuries and receive the financial compensation you deserve.
How Long Will A Medtronic Recall Lawsuit Take?
There is no one-size-fits-all answer to this question, as every case is unique, with its own unique, individual circumstances.
The individual circumstances of your lawsuit will determine how long it takes, and there are many steps to the process of filing a defective medical device lawsuit:
Hire A Defective Medical Device Lawyer
Hiring a defective medical device lawyer gives you the best chance of recovering every penny you are entitled to under California law for your injuries stemming from recalled Medtronic medical devices.
File A Complaint
The process of filing a civil lawsuit begins when your defective medical device attorney files a complaint. A complaint is a document which covers your case details, argues that a defendant is liable for your injuries, and contains the amount of money you are demanding. You typically have two years from the date of your injury to file a complaint in California.
Discovery
The discovery process comes after a complaint is filed. Both sides in the lawsuit exchange information such as medical records, company emails and product design sheets. Documents are collected, facts are unearthed, and witness statements and depositions are taken during the process of discovery. Depositions involve the two sides in a lawsuit asking each other, and witnesses, questions under oath.
Negotiations
The two sides in a lawsuit try to settle a case out of court during the negotiation phase. This phase can involve lawyers talking over the phone, mediation or arbitration. Mediation involves unbiased third parties helping the two sides in a lawsuit reach a nonbinding settlement. Arbitration involves an arbitrator deciding on a binding or nonbinding agreement.
Trial
If the two sides in a lawsuit fail to reach an agreement during negotiations, the lawsuit will move to trial. Settlements can still be reached during a trial, before a trial verdict is reached. If your HawkOne or TurboHawk Plus lawsuit goes to trial, a judge or jury will determine whether Medtronic is financially liable for your injuries.
Verdict
The judge or jury will reach a verdict, determining if Medtronic is liable for your injuries, and how much they must pay you if you win.
Appeals
The losing side in the lawsuit can appeal the verdict, and a successful appeal can lead to a new trial or a new settlement amount.
Collection
You may receive a lump sum or multiple payments once judgment collection occurs after your lawsuit is successful.
What Can I Recover In A HawkOne Or TurboHawk Plus Lawsuit?
You may be eligible to recover financial compensation for multiple types of damages in a lawsuit against Medtronic, including:
- Past and future medical bills
- Pain and suffering
- Lost wages
- Loss of earning capacity
Why Should I Sue Medtronic?
If you have suffered a serious injury after receiving a medical treatment involving a HawkOne or TurboHawk Plus device, you have probably already been left with huge medical bills regarding treatment for the injury and may require future medical care resulting from the injury that will result in even more medical bills. To make things worse, your injury may have left you unable to work for a while, further compounding your financial woes.
A lawsuit against Medtronic can help you recover all the money you have lost as a result of your injuries, but perhaps more importantly, it can hold Medtronic responsible for your injuries. Medtronic knows that its defective HawkOne and TurboHawk Plus devices can cause serious injuries such as ruptured arteries, but have refused to take these products off of the market. You can hold Medtronic accountable for this egregious decision to place profits over the safety of patients by filing a lawsuit with the help of our experienced defective medical device lawyers.
Call us today at 800-718-4658 for a free consultation if you or a loved one was injured by a HawkOne or TurboHawk Plus directional atherectomy system. You may be eligible for financial compensation.